We offer cost-effective iPSC differentiation services with fast turnaround times to support your research. Our tailored protocols cater to both small and large-scale production based on your specific needs.
Our protocols can be tailored from small to large scale production based on your needs. Upon request, our dedicated stem cell specialists will provide free consultation and ongoing support for your disease modeling studies.



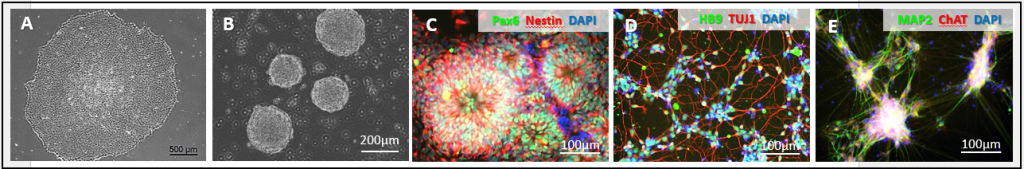
Figure 1. Human iPSC-motor neuron differentiation. (A) Phase contrast image of iPSC before differentiation. Scale bar: 500 µm; (B) Neural spheres upon neural induction on Day 11. Scale bar: 200 µm;(C) Neural rosette formation in neural progenitor cells (NPCs). Scale bar: 100 µm; (D and E) The cryopreserved iPSC-derived motor neurons were recovered and cultured 200,000 viable cells in Motor Neuron Maintenance Medium on PDL / Cultrex-coated wells in the 48-well plate; (D) The cells were fixed and stained with anti-HB9 and anti-TUJ1 on Day 2 post thaw; (E) The cells were fixed and stained with anti-MAP2 and anti-ChAT on Day 7 post thaw. Scale bar: 100 µm.



Gasset-Rosa, F., Lu, S., Yu, H., Chen, C., Melamed, Z., Guo, L., . . . Cleveland, D. W. (2019). Cytoplasmic TDP-43 de-mixing independent of stress granules drives inhibition of nuclear import, loss of nuclear TDP-43, and cell death. Neuron, 102(2). doi:10.1016/j.neuron.2019.02.038 — Learn More
Drug discovery, drug screening, disease modeling, personalized medicine, and preclinical cell regeneration projects. Our approach allows us to develop tailored solutions that address your unique needs and accelerate the path to success.
Gene editing services using CRISPR/Cas9 technology for SNP replacement, large reporter gene knock-in, and DNA knock-out in iPSC and other cell lines. Our experts have extensive experience and can work with in-house or provided cell lines.
Customized cell isolation services with an extensive network of ethically procured tissues from donors with specific requirements. Available in a variety of formats and can be obtained from a wide range of species and diseases.
Please provide the project information in the inquiry form, including but not limited to starting cells, desired differentiated cell type, scale of differentiation, specific markers of interest and any functional assay of interest.
It takes 2-3 months depending on the recovery of the cells provided by the customer, cell type to be differentiated, and the scale of differentiation.